UMN - “Un-TIL It’s Cured” Initiative
Last year, an estimated 2 million new cases of cancer were diagnosed in the US, and over 600,000 Americans died of cancer. Of the two main types of cancer (solid and blood), solid cancer is the most common, comprising 80-90% of cancer cases.
Localized solid tumors can often be treated well with surgery and radiation. But late-stage solid cancer, in which cancer has spread throughout the body, typically has a dire prognosis. Very few effective options for treatment exist. Death is likely.
Venn’s PRI to the University of Minnesota under our technology transfer funding model aims to improve a promising treatment for late-stage solid cancer using genetically engineered Tumor Infiltrating Lymphcytes (TIL).
Tumor Infiltrating Lymphoctes (TIL)
Since the 1990's, researchers have studied how to help each patient's own immune cells do a better job of fighting cancer cells.
Tumor Infiltrating Lymphocytes (TIL), often called "T-Cells," are one type of immune cell that naturally enter tumors to recognize and attack cancer cells. In late-stage cancer patients, TIL are overwhelmed and ineffective.
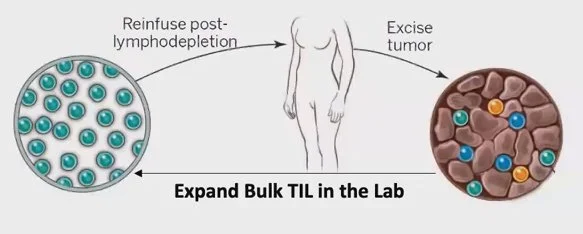
TIL Therapy
TIL Therapy involves extracting TIL from a patient's tumor, growing them to large numbers in the lab, and infusing billions of the rejuvenated TIL cells back into the patient to fight the cancer.
TIL Therapy has shown a lot of promise, eliciting favorable clinical responses for some patients and with certain types of cancer that had otherwise proven untreatable.
Over time, scientists have learned how to incrementally increase effectiveness of TIL Therapy in many ways. For example, researchers can sort through a patient's TIL to find the best type of TIL and then grow only those types. But major limitations for TIL Therapy remain.
Genetically Engineered TIL
Using powerful new genetic engineering methods like CRISPR/Cas9, it is now possible to change the genetic makeup of cells.
Over the last five years, renowned UMN researchers Dr. Branden Moriarity and Dr. Beau Webber have been genetically engineering TIL to make TIL Therapy even more effective.
One major breakthrough came from engineering patient TIL cells to remove a problematic gene (CISH) that suppresses TIL's ability to find and eradicate cancer cells.
Recently, the researchers completed a landmark Phase I clinical trial that tested their genetically engineered TIL in 12 end-stage gastro-intestinal cancer patients who had no other therapeutic options. The results were unprecedented.
Remaining Hurdles to Overcome
Given the promising results of the Phase I clinical trial, Drs. Moriarity and Webber believe they are on the verge of figuring out how to scale customized TIL Therapy.
But three major challenges remain to be overcome:
Improve TIL Function
Further genetic engineering of TIL needs to be done to improve tumor recognition and killing power, increasing the number of patients who experience a complete response.Enable Faster Manufacturing
For custom TIL Therapy to be viable in the clinic, it needs to be faster. In their Phase I trial, it averaged 123 days to deliver TIL Therapy, and several patients passed away before it was finished. The team has already identified ways to reduce this period to 20-25 days.Reduce Cost
Each custom TIL Therapy in the trial cost $500,000+ per patient to manufacture. Reducing these costs will help speed up research as well as make TIL Therapy more affordable and accessible.
Recipient Name: University of Minnesota Technology Commercialization
Amount Raised: $400,000
Investment Sought: Up to $2 million
Overview of Investment Terms:
Variation of Recoverable Grant
If the University earns revenue from the license, the University will split it with Venn on a 50/50 basis up until a maximum 3X return of the original Venn funding.